In the intricate dance of cellular communication, receptors play a crucial role as the sentinels of the cell membrane, poised to detect and respond to incoming signals from the external environment. Among these, G-protein-coupled receptors GPCRs stand out as key players, orchestrating a cascade of events upon activation by specific signal molecules. Now, a groundbreaking study led by an international team of researchers, with significant contributions from Friedrich-Alexander University Erlangen-Nürnberg FAU, has unveiled the intricate molecular choreography underlying the activation of an important GPCR, offering unprecedented insights into the mechanisms governing cellular signaling.
Published in the esteemed journal Nature, the research harnesses the power of time-resolved cryogenic electron microscopy to “film” the activation of the β2-adrenergic receptor, a pivotal GPCR involved in regulating heart and lung function. Led by Georgios Skiniotis from Stanford University, the collaborative effort brings together a multidisciplinary team of scientists, including Prof. Dr. Peter Gmeiner from FAU, Brian Kobilka from Stanford University, and Peter Hildebrand from Universität Leipzig and Charité Berlin.
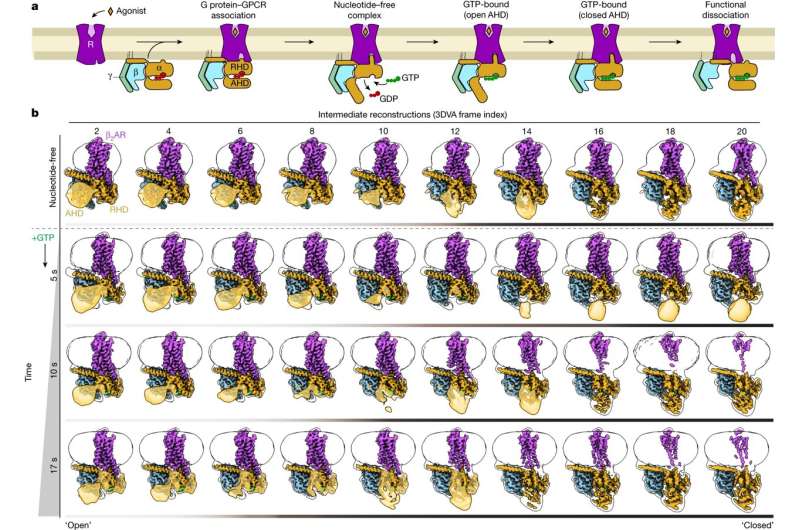
At the heart of the study lies the innovative use of cryo-electron microscopy, a cutting-edge technique that enables researchers to visualize molecular structures with unprecedented clarity. By rapidly freezing the receptor-G-protein complex at different time points following activation, the researchers were able to capture a series of snapshots depicting the dynamic conformational changes undergone by the receptor upon binding to its agonist molecule.
“For the first time, we were able to observe the activation process of the β2-adrenergic receptor at the atomic level,” explains Prof. Gmeiner, highlighting the significance of the findings. “By dissecting the complex interplay between the receptor and the G-protein, we gained invaluable insights into the molecular mechanisms driving cellular signaling.”
Central to the success of the study was the development of a novel “super-adrenaline” molecule by Prof. Gmeiner’s group at FAU. This potent agonist binds tightly to the β2-adrenergic receptor, stabilizing the receptor-G-protein complex and enabling the visualization of conformational changes without the need for adapter proteins. The enhanced stability afforded by the super-adrenaline molecule allowed the researchers to elucidate the intricate dynamics of G-protein activation with unprecedented clarity.
“Our findings pave the way for the development of more effective drugs targeting GPCRs, with potential applications in treating a wide range of diseases,” remarks Prof. Gmeiner, underscoring the translational implications of the study. GPCRs represent attractive drug targets due to their central role in cellular signaling, and the insights gleaned from this research hold promise for the development of precision medicines with enhanced efficacy and reduced side effects.
Indeed, the Nobel Prize-winning work of Brian Kobilka, who pioneered the structural elucidation of GPCRs using X-ray crystallography, serves as a testament to the importance of understanding the molecular basis of receptor activation. By unraveling the intricate dance of GPCR signaling, researchers are poised to unlock new avenues for drug discovery and therapeutic intervention.
As the field of structural biology continues to advance, driven by technological innovations such as cryo-electron microscopy, the quest to decipher the molecular mysteries of life’s fundamental processes enters a new era of discovery. With each groundbreaking revelation, we inch closer to unraveling the intricate tapestry of cellular signaling and harnessing its potential for the benefit of human health and well-being.