In the realm of genetics, the intricate dance between nature and nurture plays out on a molecular stage, shaping the destiny of every cell in our bodies. At the heart of this intricate process lies epigenetics – the silent conductor orchestrating the symphony of gene expression without altering the underlying DNA sequence.
Recent groundbreaking research, led by scientists at the University of Hong Kong, has unveiled the secrets of epigenetic inheritance, shedding light on the inner workings of the DNA copying machinery and its pivotal role in passing on gene traits from one generation to the next.
Published in the esteemed journal Nature, the study marks a significant milestone in our understanding of epigenetics – a field poised to revolutionize our approach to healthcare and disease prevention.
In the intricate tapestry of life, every cell contains the same genetic blueprint encoded within its DNA. Yet, remarkably, cells exhibit diverse functions and identities, governed by the complex interplay of epigenetic marks that adorn the chromatin landscape.
Epigenetic modifications, such as histone acetylation and DNA methylation, serve as molecular switches, toggling genes on or off in response to environmental cues. This dynamic regulation of gene expression underpins cellular differentiation, development, and adaptation to changing conditions.
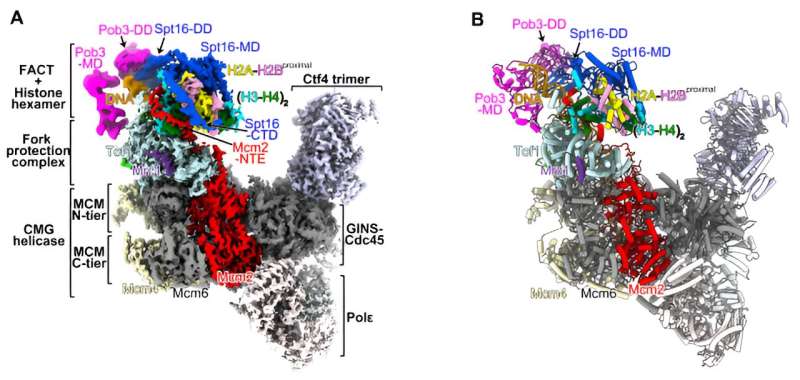
Central to the preservation of epigenetic information is the DNA copying machinery, known as the replisome. Like a diligent custodian, the replisome ensures the faithful transmission of epigenetic marks during the frenzied chaos of cell division.
Through a delicate choreography of protein interactions, the replisome dismantles parental nucleosomes – the building blocks of chromatin – and faithfully replicates their epigenetic signatures onto newly synthesized DNA strands. This meticulous process ensures that each daughter cell inherits the same epigenetic landscape as its parent.
The University of Hong Kong research team employed state-of-the-art cryo-electron microscopy techniques to capture detailed snapshots of the replisome in action. By visualizing the intricate molecular structures of the replisome complex, they unraveled the mechanisms underlying epigenetic inheritance at a molecular level.
Their findings revealed the dynamic interplay between the replisome, histones, and accessory proteins, providing unprecedented insights into the molecular machinery responsible for maintaining epigenetic stability.
The implications of this research are far-reaching, with profound implications for human health and disease. Epigenetic dysregulation has been implicated in a myriad of pathological conditions, including cancer, neurodegenerative disorders, and aging.
By deciphering the molecular mechanisms of epigenetic inheritance, researchers may unlock new therapeutic strategies for combating these complex diseases. Targeted interventions aimed at restoring epigenetic balance could hold the key to personalized medicine and precision healthcare.
The study underscores the intricate interplay between genetic and environmental factors in shaping cellular identity. Epigenetic modifications serve as a molecular record of our life experiences, reflecting the impact of nutrition, stress, and lifestyle on gene expression.
As we peer into the molecular depths of epigenetic inheritance, we embark on a journey towards a future of precision healthcare. By deciphering the language of epigenetics, researchers are poised to unravel the mysteries of human health and disease, paving the way for transformative advances in medicine.
The University of Hong Kong research represents a beacon of hope in this quest for knowledge, illuminating the path towards a deeper understanding of the fundamental mechanisms governing life itself. As we continue to unlock the mysteries of epigenetic inheritance, we inch closer to a future where personalized treatments and interventions hold the promise of a healthier, more vibrant world.