In a collaborative effort between Yale University and the Department of Energy’s Oak Ridge National Laboratory researchers have uncovered groundbreaking insights into addressing a significant challenge in industrial operations—silica scaling in water treatment systems. By combining experiments with molecular dynamics simulations, the team has shed light on the mechanisms underlying silica scale inhibition, paving the way for more efficient and sustainable water treatment processes.
Silica scaling, a prevalent issue in industrial water treatment systems, occurs when dissolved silica precipitates out, forming solid deposits that can impair equipment performance and longevity. These deposits not only increase maintenance costs but also pose the risk of system failures. Conventional methods of combating silica scaling, such as adjusting the pH of water, are costly and often exacerbate other forms of scaling.
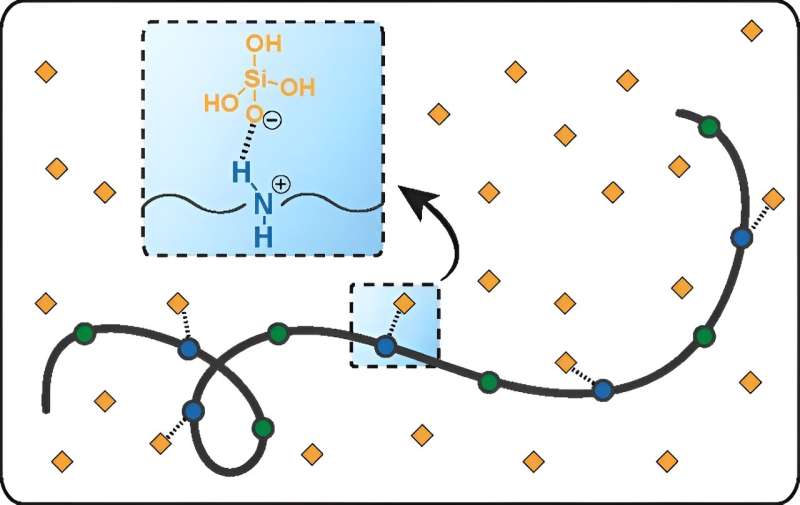
To address this challenge, researchers have turned to silica-inhibiting polymers, or antiscalants, as a potential solution. However, the mechanisms by which these antiscalants work and how to enhance their effectiveness have remained poorly understood. The collaborative study sought to unravel these mysteries and explore the molecular design of functional polymers for silica scale inhibition.
By synthesizing a series of nitrogen-containing polymers and testing their performance in oversaturated silicic acid solutions, the researchers discovered significant variations in effectiveness among similar types of antiscalants. Through molecular dynamics simulations conducted at ORNL, the team gained insights into the specific physical and chemical properties of these polymers, elucidating the mechanisms underlying silica scale inhibition.
The findings, detailed in a paper titled “Molecular Design of Functional Polymers for Silica Scale Inhibition,” published in Environmental Science & Technology, highlight the critical role of charged amine and uncharged amide groups in polymer backbones for superior silica scale inhibition performance. The simulations revealed strong binding between deprotonated silicic acid and polymers with protonated amine groups, shedding light on the synergistic contributions of functional groups in the scale inhibition process.
Importantly, the research offers a deeper understanding of silica scaling phenomena and provides a roadmap for the development of more effective antiscalant solutions. While current efforts focus on addressing silica scaling in water treatment processes, the ultimate goal is to discover universal antiscalants capable of inhibiting all types of scale formation. The molecular insights gained from this study will guide future research endeavors toward achieving this ambitious objective.
The collaborative nature of the research underscores the importance of interdisciplinary collaboration in tackling complex scientific challenges. By leveraging expertise from multiple disciplines, the scientific community can accelerate the development of innovative solutions and pave the way for more sustainable industrial practices.
Through continued research and collaboration, scientists aim to translate these findings into practical applications, ultimately revolutionizing water treatment processes and enhancing the efficiency and sustainability of industrial operations.