In the pursuit of sustainability, finding environmentally friendly alternatives to traditional chemical synthesis processes is crucial. Acetylene (C2H2), a versatile hydrocarbon with diverse industrial applications, presents a significant challenge due to its conventional production methods’ high carbon emissions. However, a groundbreaking collaboration between Doshisha University and Daikin Industries, Ltd., Japan, has introduced a novel approach to synthesize acetylene using carbon dioxide (CO2) and water (H2O) as raw materials. Published in ACS Sustainable Chemistry & Engineering, their research signifies a significant leap towards achieving carbon neutrality and building sustainable societies.
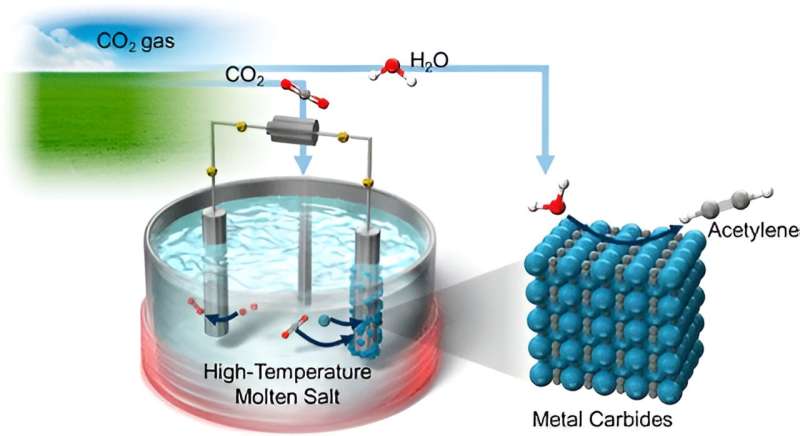
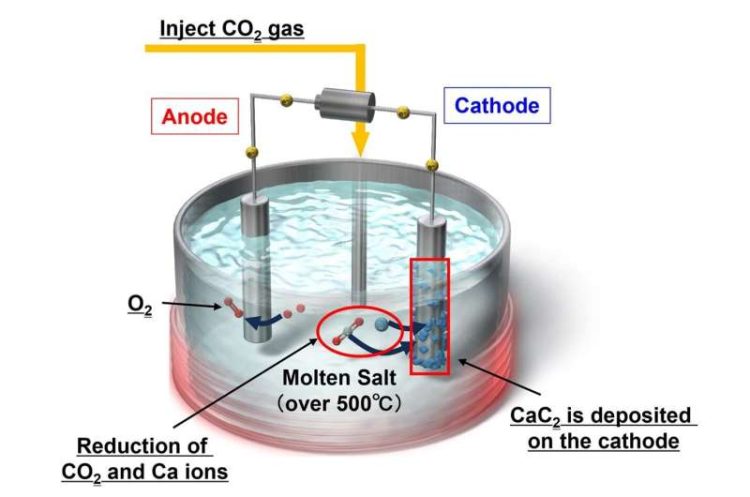
The Innovative Synthesis Approach: The research team’s strategy revolves around the electrochemical and chemical conversion of CO2 into acetylene, facilitated by high-temperature molten salts, particularly chloride melts. At the heart of the process lies the utilization of metal carbides, solid compounds composed of carbon and metal atoms, as catalysts for the conversion. Dr. Yuta Suzuki, Assistant Professor at Harris Science Research Institute, elucidates the process, wherein CO2 is initially transformed into metallic carbides like CaC2 and Li2C2, which then react with H2O to yield C2H2 gas.
Optimization and Efficiency: To enhance energy efficiency and efficacy, the researchers conducted a series of experiments exploring various electrode materials and molten salt compositions. Through meticulous analysis, including cyclic voltammetry and X-ray diffraction, they identified a NaCl−KCl−CaCl2−CaO melt saturated with additional CaCl2 in a CO2 atmosphere as the optimal configuration. This specific melt facilitated the selective formation of CaC2 around the cathode, showcasing superior performance compared to other compositions.
Advantages and Potential Applications: The proposed approach offers several notable advantages over conventional synthesis pathways for acetylene. Firstly, the ability to reuse electrodes following a simple reconditioning process underscores the method’s sustainability and cost-effectiveness. Moreover, by utilizing CO2 as a feedstock, the process contributes to carbon neutrality and mitigates reliance on fossil fuels. Prof. Takuya Goto emphasizes the technique’s potential as a carbon-negative emission technology, particularly when combined with direct air capture processes. This visionary approach opens doors to sustainable resource utilization and energy cycles, aligning with the imperative of reducing environmental impact.
Continued research and development of this innovative method hold immense promise for transforming the landscape of chemical synthesis and fostering sustainable practices across industries. As efforts progress, the scalability and commercial viability of the process are expected to improve, driving widespread adoption and impact. Ultimately, harnessing carbon dioxide and water to produce acetylene represents a paradigm shift towards achieving harmony between industrial progress and environmental stewardship. By embracing innovative solutions, we can pave the way towards a sustainable future while preserving the essence of modern civilization.